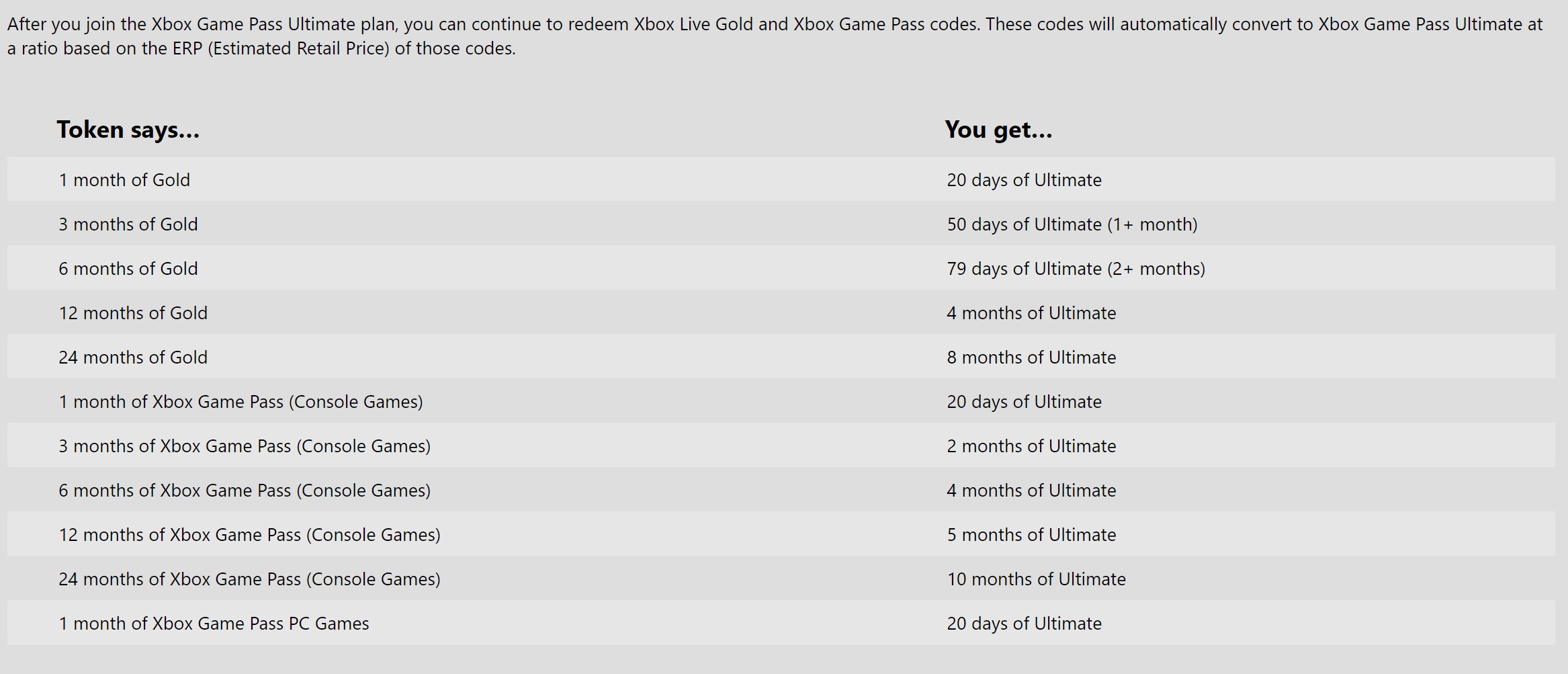
La Increíble Historia Del Empleado De Microsoft Que Amasó Una Fortuna De Más De 10 Millones De Dólares Robando Tarjetas Regalo De Xbox | huntingtonchiropractor.com

7-Eleven on Twitter: "Tenemos las tarjetas Xbox Live Gold de 12 meses al 25% de descuento. ¡Aprovecha! http://t.co/Gruyj3pFwl" / Twitter