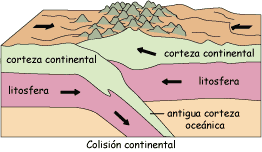
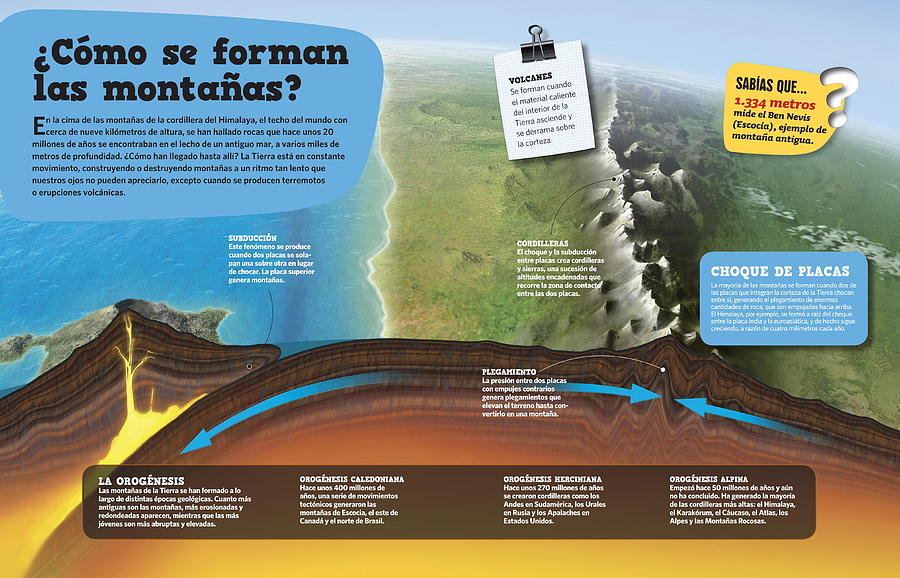
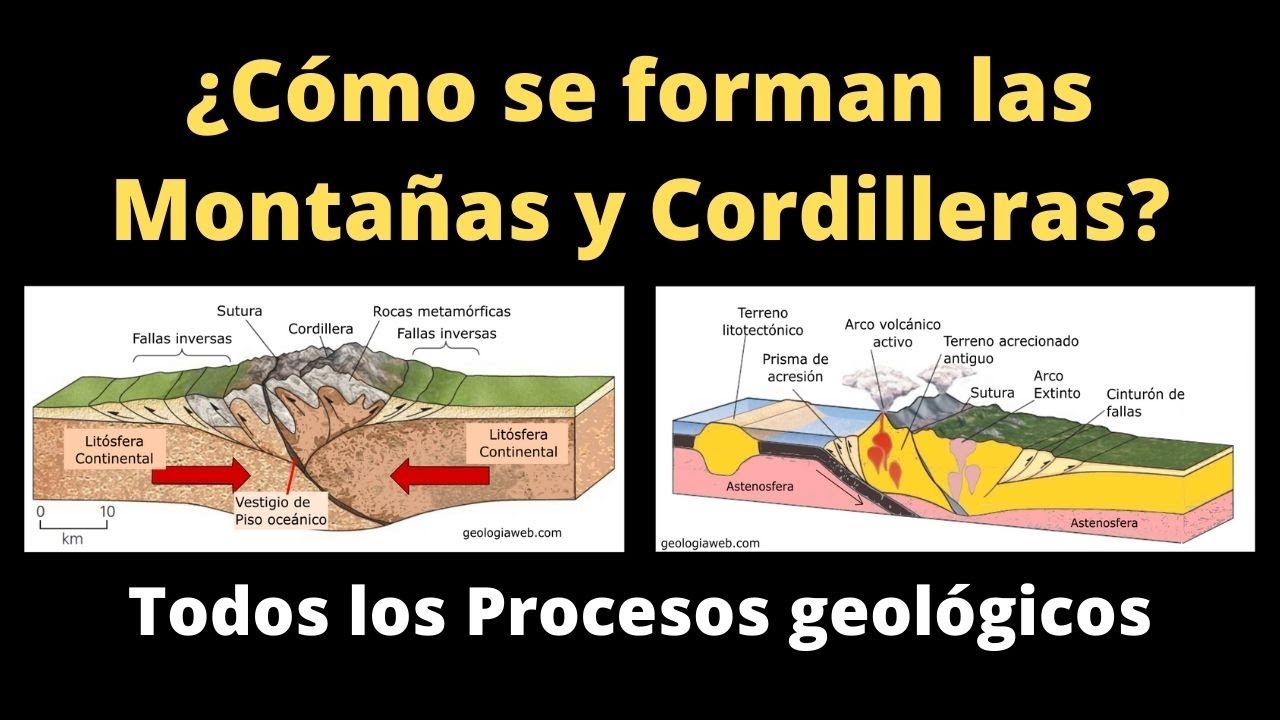
Un Hermoso Paisaje De Campos En Las Montañas De Almora. Una Visión De Cómo Se Hace La Agricultura En Terraza En Uttrakhand Imagen de archivo - Imagen de arroz, paisaje: 195310809

Un Hermoso Paisaje De Campos En Las Montañas De Almora. Una Visión De Cómo Se Hace La Agricultura En Terraza En Uttrakhand Imagen de archivo - Imagen de hermoso, planta: 193264657

Cómo hacer las montañas en una maqueta de tren | Trenes de juguete, Como hacer maquetas, Mesa de tren