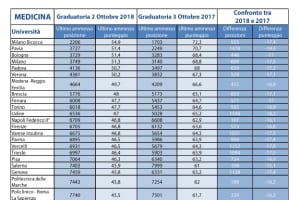
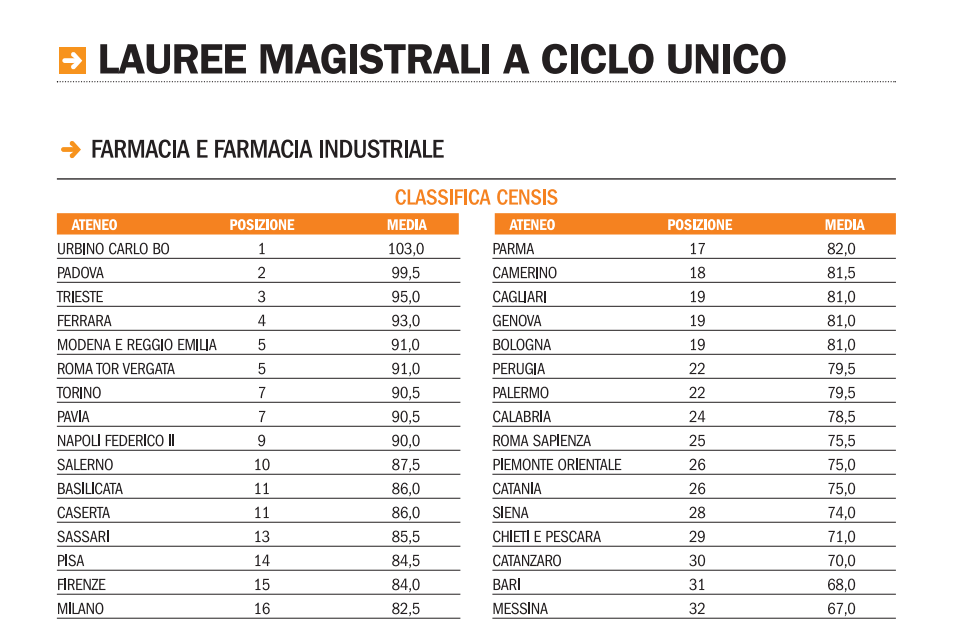
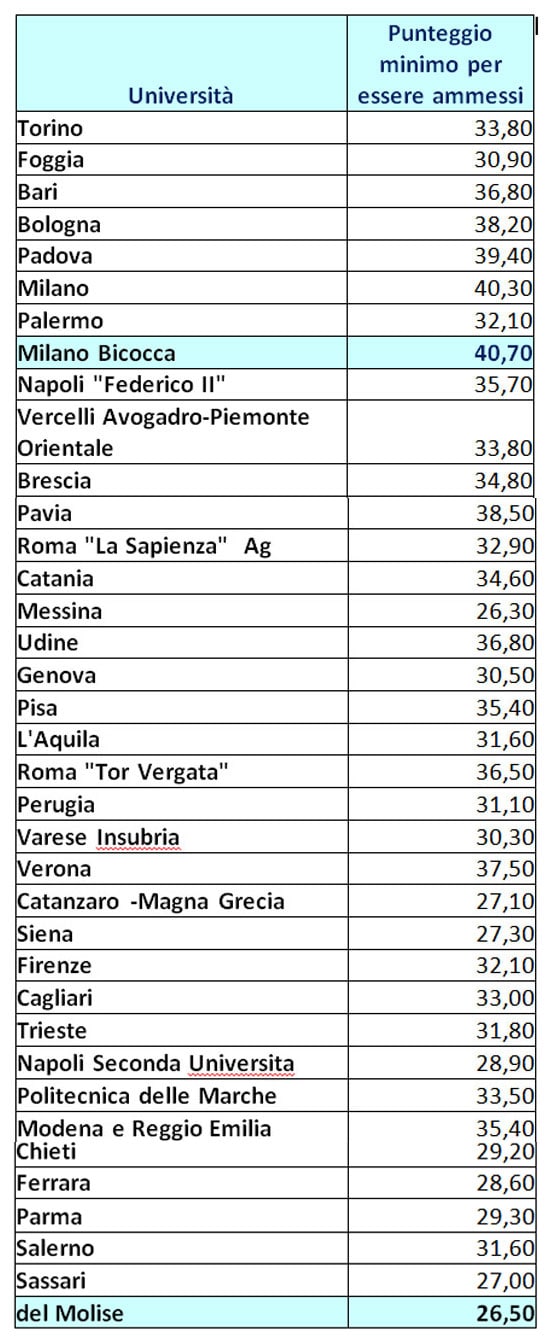
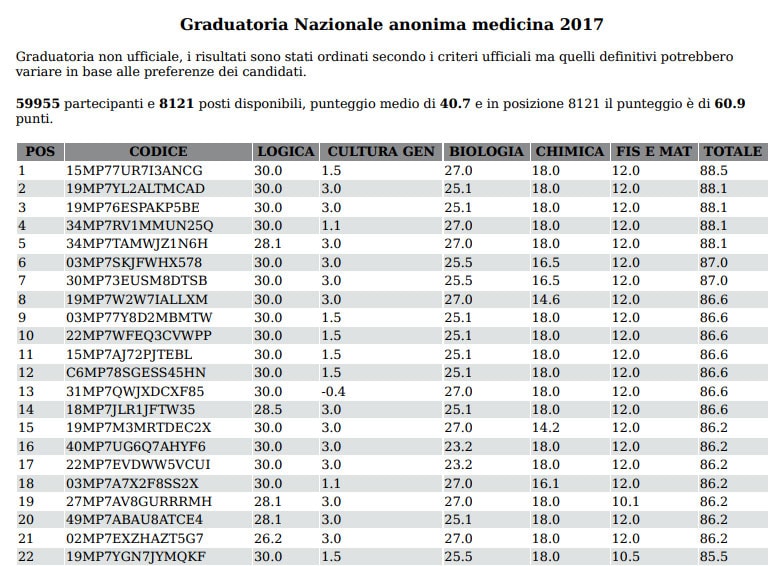
Immissioni in ruolo 2020/21, elenchi aggiornati GaE: dati ufficiali da tutti gli USR (aggiornamento 25 agosto) - ScuolaInforma
GRADUATORIA ALTRE PROFESSIONALITA SANITARIE PSICOLOGI SPECIALIZZAZIONE PSICOLOGIA - ASST DI PAVIA - SEDE DEL COMITATO CONSULTIVO