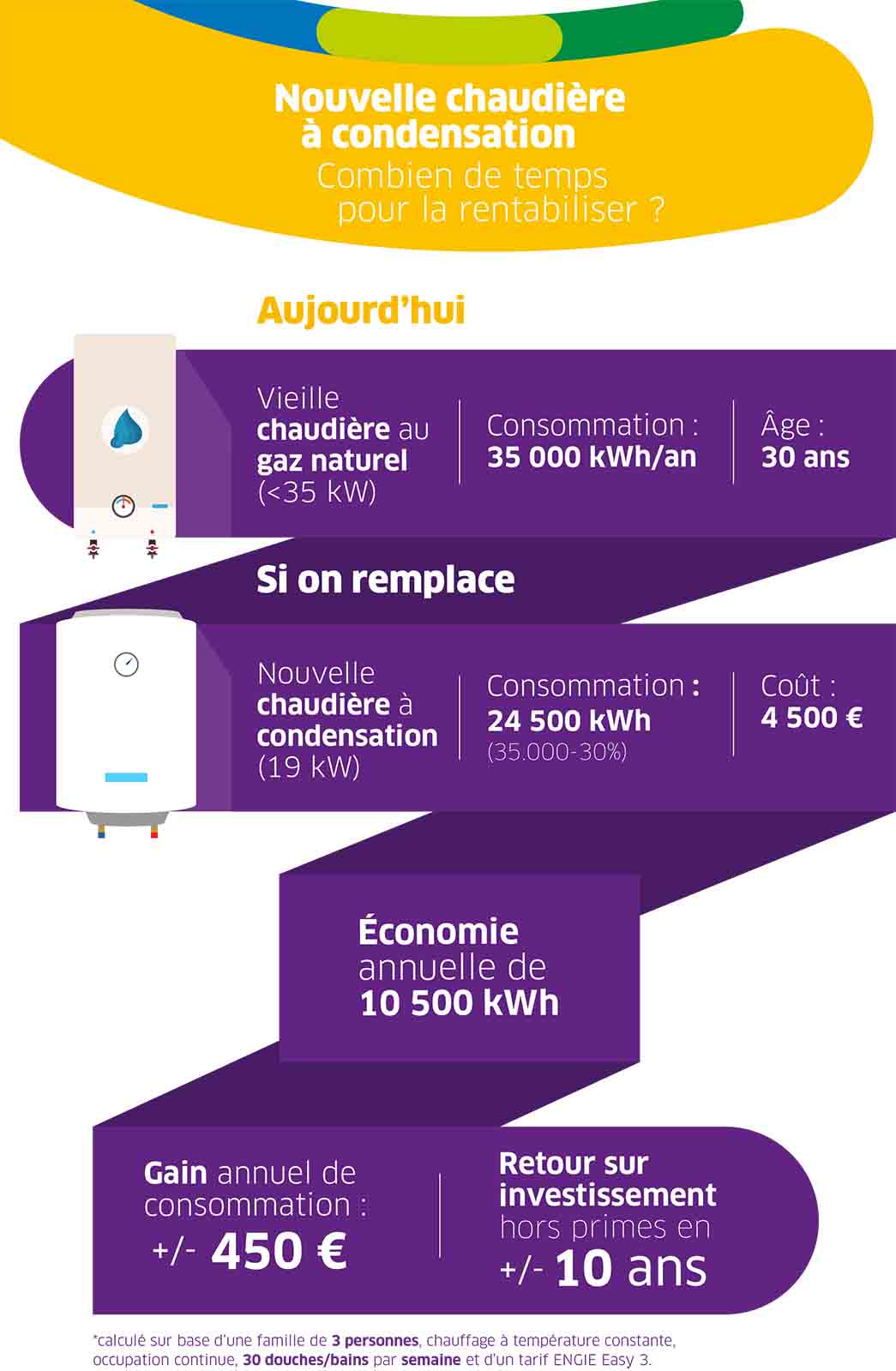
Le Parisien - L'installation de chaudières au fioul, trop polluantes, sera interdite à partir du 1er janvier 2022. Des aides peuvent alléger votre facture si vous devez changer de système de chauffage >
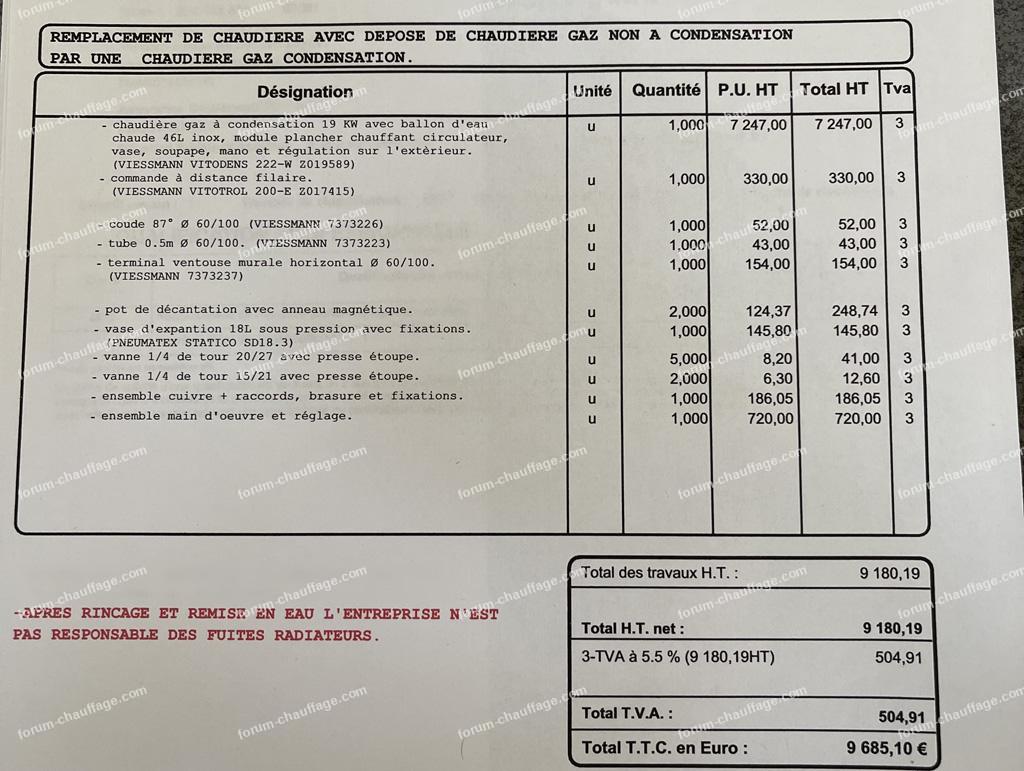
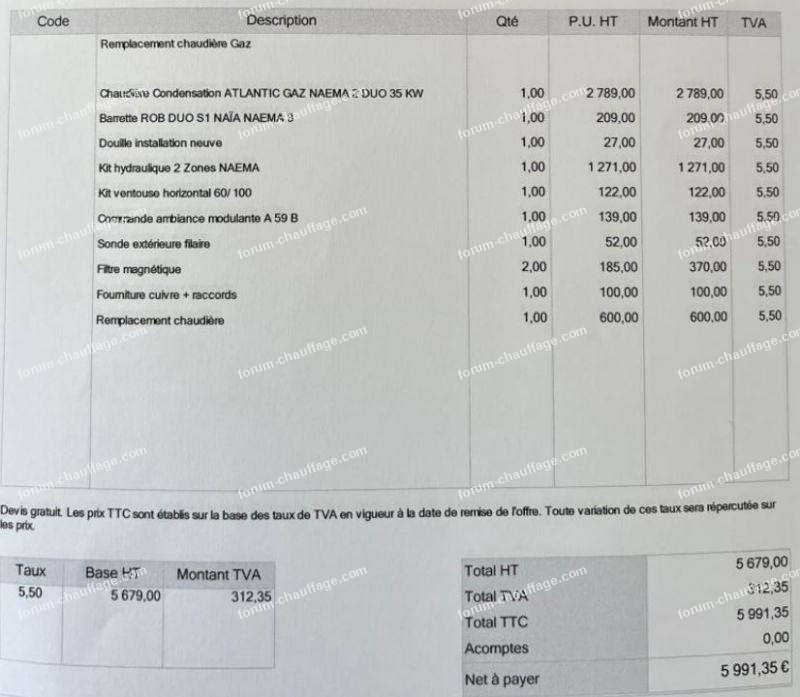
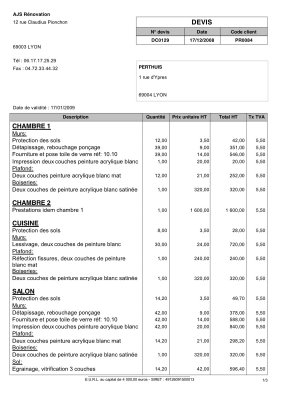
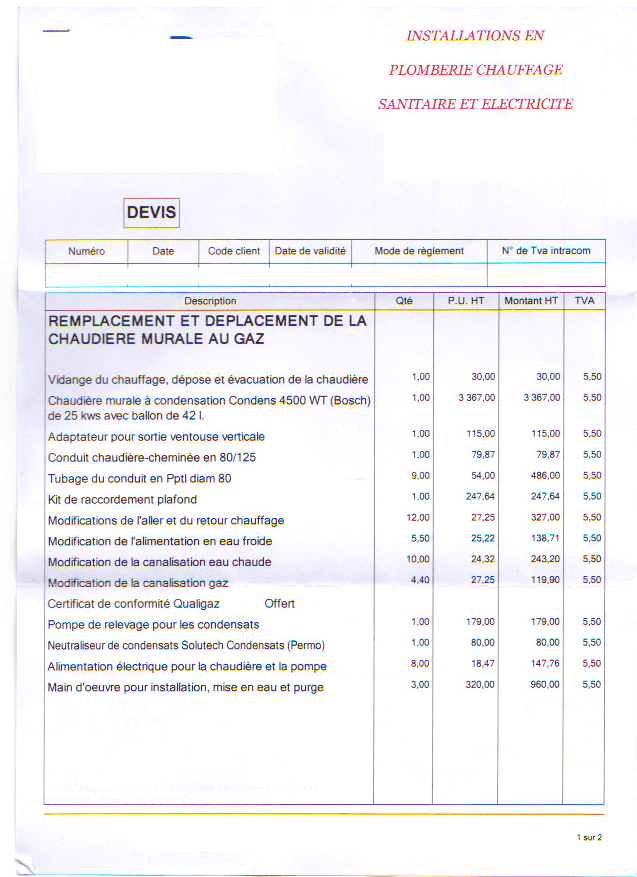
Avis sur devis de Chaudière à Gaz différence de prix colossale | Questions / Réponses Forum Chauffage